 The Chemistry of Natural Waters
The Chemistry of Natural Waters 
The Meaning of Water Hardness
The hardness of water is a property of water in which it forms an insoluble curd with soap (soap scum) instead of foaming lather. This property of water is due to the presence of any chemical in water, such as Magnesium, calcium, chloride, sulfate and bicarbonate ions. Water hardness is a measurement of dissolved calcium (Ca2+) and magnesium (Mg2+) in water. The more Ca2+ ions and Mg2+ ions in water, the harder the water. When hard water is boiled, there may remain white residues after all water has evaporated (2).
Importance of Water
Water is one of the necessities to human. Every person needs at least 80 ounces of water per day to maintain a healthy condition. Therefore, the quality of drinking water is important. In this case, the water hardness directly influences people because if the water is too soft, it means people are not able to get enough amount of ions they need (1).
Advantages of Water Hardness
Hard water has some health benefits, which include the fact that it can reduce cases of hypertension and tetany. Besides, hard water also reduces the prevalence of arteriosclerotic heart diseases, sudden death resulting from cardiac arrest, tetany, or hypertension. Finally, it is also important in that it prevents degenerative heart diseases.
How to Measure Water Hardness
There are various scales, which are used to measure water hardness all over the world. These scales include British degree, American degree, French degree and German degree. Water hardness expressed in degrees that are grain per gallon. The degree as a unit of measuring the hardness of water is the measure of grains of Calcium Carbonate that is present in 70, 000 grains of water. Besides, it can also be expressed by Parts Per Millions (PPM). PPM is the quantity of the number of grains of calcium carbonate that is present in one million grains of water.
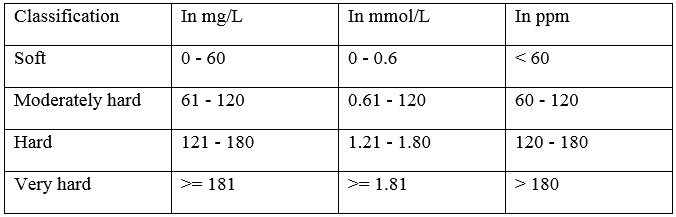
According to varying amount of Ca2+ and Mg2+ ions dissolved in water, water hardness could be separated into four categories, which include are soft, moderately hard, hard, and very hard. To determine the hardness of water, there are several units to use, such as Molarity (M), Parts Per Million (ppm) of hardness (CaCO3), and the ppm of ions (Ca2+ and Mg2+). From Table 1, readers can understand the range of different categories of waters. (3)
Table 1. General Guidelines for Classification of Hardness
Methods of Determining Water Hardness
There are several techniques to determine water hardness. The Total Dissolved Solids (TDS) test involves analysis of the residue. The residue is attained by boiling sample water so as to make it evaporate and remain with the residue. Besides, EDTA and AA are two other main techniques to determine water hardness.
Determining Water Hardness Using EDTA Method
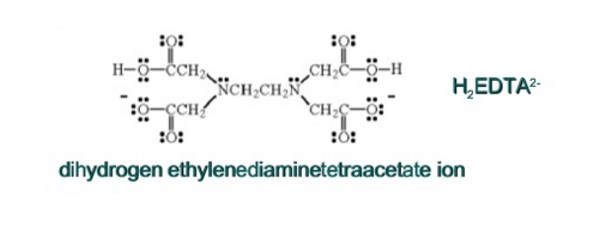
It is important to reiterate that water is said to be ‘hard’ when it contains Mg2+ and Ca2+ ions. They are the ions, which react with soap to form an insoluble white substance called ‘soap scum.’ The above two ions together with others such as Pb2+ and Fe2+ can be removed from a sample hard water by adding ethylenediaminetetraacetic acid (EDTA). This chemical reaction that takes place its mad4e possible by the fact that EDTA has a greater affinity for Mg2+ and Ca2+, especially, when it is in the form of the dihydrogen anions H2 EDTA2-, which is the ionic form of EDTA at pH 10.
It is important to reiterate that water is said to be ‘hard’ when it contains Mg2+ and Ca2+ ions. They are the ions, which react with soap to form an insoluble white substance called ‘soap scum.’ The above two ions together with others such as Pb2+ and Fe2+ can be removed from a sample hard water by adding ethylenediaminetetraacetic acid (EDTA). This chemical reaction that takes place its mad4e possible by the fact that EDTA has a greater affinity for Mg2+ and Ca2+, especially, when it is in the form of the dihydrogen anions H2 EDTA2-, which is the ionic form of EDTA at pH 10.
“EDTA, ethylene EDTA, ethylenediaminetetraacetic acid, has four carboxyl groups and two amine groups, which can act as electron pair donors, or Lewis bases. The cability of EDTA to potentially donate its six lone pairs of electrons for the formation of coordinate covalent bonds with metal cations makes EDTA a hexadentate ligand.” (5) EDTA forms ions with metal cations in the solution.
H2 EDTA2- reacts with a Ca2+ thereby forming ions by forming four different covalent bonds, which are referred to as coordinate covalent bonds, as shown in below.
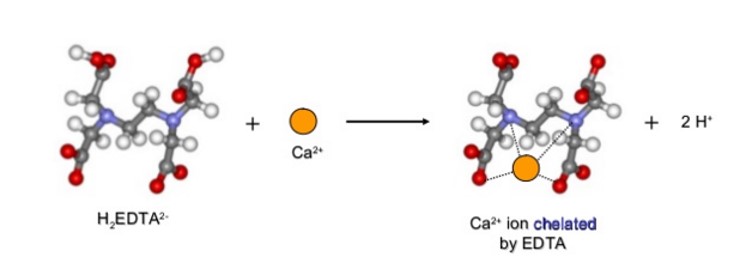
It is imperative to note that it is possible to determine the total concentration of Mg2+ and Ca2+ in a sample of hard water using EDTA in a solution buffered to a pH of 10.
Procedure of Determining Water Hardness Using EDTA Method
Firstly, use a buffer solution to adjust the pH level of water samples to reach pH 10 (base) by mixing the NH3 or NH4 solution. Then add the EBT indicator to the solution to form the HD(2-), which will make the color of solution blue. In the experiment add EBT into a solution contains magnesium ion, the solution turns red. The reason is that Mg2+ can react with EBT indicator to form MgD- ions. To find the end point, the water sample must contain Mg2+ ions before the experiment. The equation for the step is: HD(2-) (Blue) + Mg2+ → MgD(-) (Red) + H+ + Ca2+. The Ca2+ does not react with EBT indicator. After adding EDTA to the solution, it reacts with Calcium ions immediately produce CaEDTA which EDTA can always react with Mg2+ to form MgEDTA. Since Mg2+ ions formed the MgEDTA with EDTA, the HD (2-) will be formed in the solution, then change the color from red to blue. The equation for the step is: MgD (-) + H+ Ca2+ (add EDTA) → CaEDTA + MgEDTA + HD (2-) (Blue). People can find the end point of EDTA titration because the color will eventually change.
Determining Water Hardness Using Atomic Absorption Spectrophotometry (AA) Method
For another method, AA test determines the degree of different ions and atoms in sample solutions, which can absorb the light’s wavelength to characterize the atoms. The energy requires matching equally the energy, which needs to separate two electronic energy levels. When the voltage across the electrodes excites the Mg or Ca inside the lamp, the monochromatic light can be absorbed by the ions from water. Since the ions absorb the light from the lamp, it shows the concentration of the ions (6). When measuring the value by AA, it is necessary to use standard solutions, which are known for theira1a1 concentration. Then, AA will record absorbance of solutions to build a relationship between each calculated number. Therefore, when AA is measuring water sample, it will determine a precision value according to previous calculation then show on the screen.
Both EDTA titration method and AA test can use to determine the ions contain in water samples. However, EDTA titration method only roughly measures the concentration of Ca2+ ions and Mg2+ ions in the solution. The AA test can measure the specific concentration of each specific ions.
Why these Two Separate Methods are Used
People choose to use both methods by taking the advantages of the methods. The AA test can generate accurate data only when the machine is working properly. In this case, people can use the EDTA titration method to check the data generated from EDTA is accurate and in the calculated range from EDTA titration method.
Water Softening
Water softening is a chemical process that involves the partial or complete removal of the ions that cause hardness, which include Mg2+ and Ca2+. To soften the water, there are several soften substance can be added. For instance, detergents or washing soda can make water become softer results from reacting with Calcium ions to form precipitation, which can be removed by filtering process (7). Another example is Cation Exchange Resin also to remove Calcium ions from water. Most cation exchange resins contain monovalent cations, such as Na+ and or H+, which can be exchanged with the Calcium ions in water.
About the Project and the Samples Involved
Our group members decided to examine the four types of water in daily life. Jay Yang gets his bottle water which is production of Poland Spring company. Tianyi Xie gets his fountain water in campus, Penn State University, State College, PA 16803. Xuelun Yan gets his rain water also in campus, Penn State University, State College, PA 16803. I get my sample tap water at 413 East Waring Avenue, State College, PA 16801.
Hypothesis
The group thinks the water sample took from different places in Pennsylvania should have the highest hardness of water because there are mountains within the surrounding, which spreading widely in Pennsylvania. Furthermore, water gains hardness by flowing throw rocks and stones.
Procedure(4)
The PSU Chemtrek demonstrates every step of different types of experiments. In Section A, there are two separated but same water samples are used for AA to get the light absorbance data to calculate the hardness of the sample water. The water samples may be diluted with 50% pure water if the instructor recommends the same. Furthermore, the water sample may be filtered if it is cloudy, and/or contains any visible particles.
In Section B, drip one drop of distilled water, one drop of the water sample, and one drop of 1×10-3 M Ca2+ solution on a small piece of aluminum foil on the hot plate. The hot plate will evaporate the drops of solution and water sample, which the residues will leave on the aluminum foil.
In Section C and D, EDTA titration is used in the water sample. In Section C Part A, firstly, fill in one drop of EBT to first three wells. Then, add NH3/NH4Cl buffer solution to the second and third well. Lastly, add one drop of 10-3 M Mg2+ solution to the EBT/buffer solution to the third well. In Section C Part B, use a small drop pipette 6 to fill all 12 wells with one drop of 1×103 M Ca2+ solution in each well. Add one drop of EBT solution in each well. Then, add one drop of NH3/NH4Cl/MgEDTA buffer to each well. Finally, add one drop of EDTA to the first well, add two drops of EDTA to the second well, and three drops to the third wall, and then increase one drop of EDTA each time to the rest of the wells. The first well, which solution turns blue means there are excess EDTA in the well also the end point of the titration. By using the formula M (EDTA) × V (EDTA) =M (Ca2+) × V (Ca2+), the concentration of the Ca2+ can be calculated. Using the same methods, people can calculate the concentrations of Mg2+ and Ca2+.
In Section E and F, the experiment demonstrates two methods to soften water. The first method is by adding 20 mg of commercial conditioning product to the water sample and mix them up. Then, repeat the steps from the previous part of the EDTA titration test to the new water sample. Last, examine the sample water from a specific well first becomes blue and compare it with the normal water sample. The second method is by exchanging ions. Add the ion exchange chemicals to the water sample instead of the commercial conditioning product then do the same EDTA titration test. Then, compare the number of the wells (the first well, which turns blue) with the normal water sample (4).
Results (8) (9) (10) (11)
Table 2. Observation from TDS tests
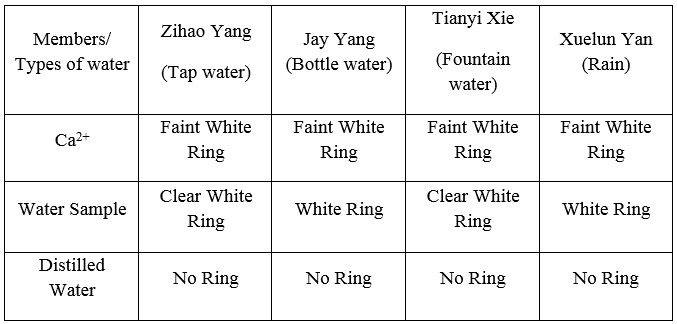
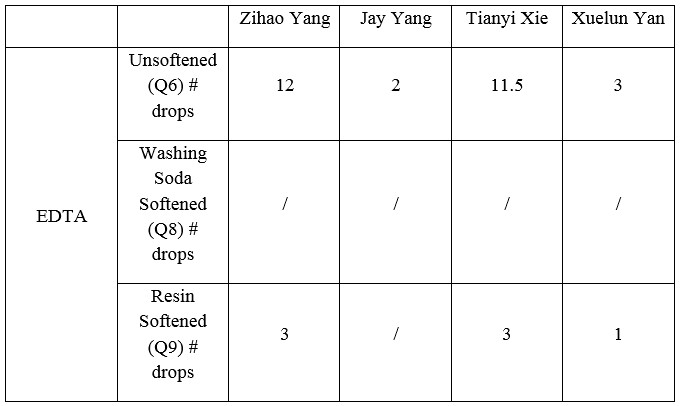
Table 3. Drops of EDTA with water samples
Sample Calculation: (Zihao Yang’s Result)
- Molarity Zihao Yang’s sample:
M(EDTA) × V(EDTA) = M(Sample) × V(Sample)
(2.00 × 10-4 M) (4 drops) = (M (Sample)) (1 drop)
M(Sample) = 8.00 × 10-4 M
- Parts Per Million (ppm) of CaCO3
Calculation Formula: (1×10-3 moles CaCO3/ 1 liter of solution) (100.0 g CaCO3/ 1 mole CaCO3) (1000mg CaCO3/ 1 g CaCO3) = 100 ppm
Zihao Yang’s Sample: (8.00 × 10-4 M)
(8.00 × 10-4 M)(100.0 g CaCO3/ 1 mole CaCO3)(1000mg CaCO3/ 1 g CaCO3) = 80 ppm
Table 4. Different Units of Concentration of Unsoftened Water
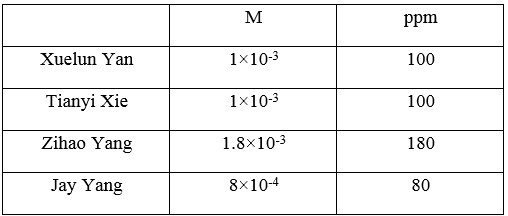
Table 5. AA Standards
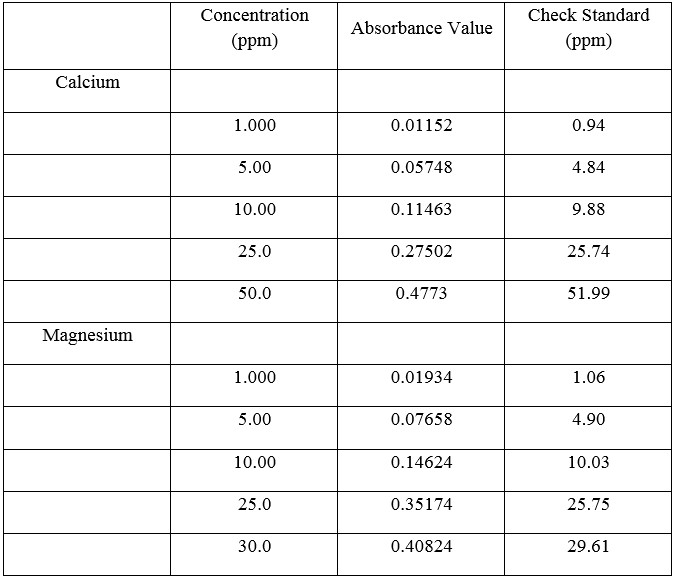
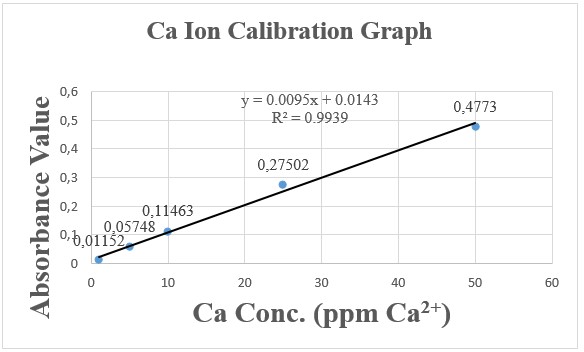
Graph 1 and Graph 2 are based on the data given in Table 5
Graph 1: Absorbance Value vs. Concentration of Ca2+
Graph 2: Absorbance Value vs. Concentration of Mg2+

Table 6. AA absorbance test
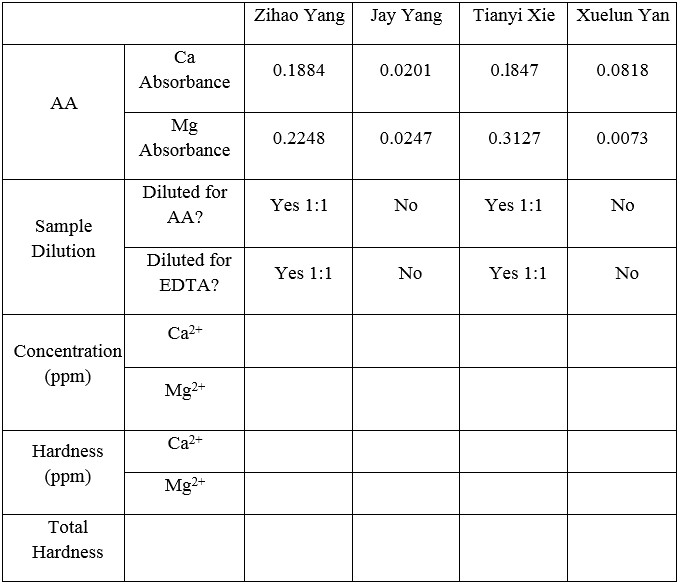
Use AA absorbance data and equation to calculate the water sample hardness:
Ca equation: y = 0.0095x + 0.00143 (y = AA absorbance data, therefore, plug in y value to solve for x, which is the amount ppm for Ca2+)
0.1767 = 0.0095x + 0.00143 x = 18.449 ppm Ca2+
Mg equation: y = 0.0135x + 0.088 (y = AA absorbance data, therefore, plug in y value to solve for x, which is the amount ppm for Mg2+)
0.1799 = 0.0135x + 0.0088 x = 12.674 ppm Mg2+
(26.625 ppm Ca2+)[100g/mol (CaCO3)]/[40.0g/mol (Ca2+)] = 66.6 ppm CaCO3
(13.448 ppm Mg2+)[100g/mol (CaCO3)]/[24.3g/mol (Mg2+)] = 55.4 ppm CaCO3
The Total Hardness of the water sample by AA: 66.6 ppm + 55.4 ppm = 122 ppm
Discussion
For the ETD test (Table 2), all of our group members have white rings and even darker colored ring in the test, which indicates that all of our waters samples have more Ca2+ because the residues are clearer than what remains from the 1×103 M Ca2+ solution. In our ETD test, the 1×103 M Ca2+ solution does not show clear rings from Carol, Chen and Adam Holyk’s report and only faint white ring report by Jason Cillo and Bo-Yang Chen.
In the EDTA test (Table 3 and Table 4), Bo-Yang Chen’s water sample from California has the highest concentration of the ions, which is 180 ppm hardness. And, the water sample from McDonald, Pa has the lowest 80 ppm hardness. The two remain water samples have the same 100 ppm of hardness. In the AA test (Table 6), Bo-Yang Chen and Jason Cillo both diluted the sample water with 50% of pure water. From Bo-Yang Chen’s water sample, the total harness of water is 122 ppm, which is higher than other group members. By comparing the AA test data with EDTA titration test, we can tell Bo-Yang Chen has the highest hardness of water hardness among other three members.
After using the baking soda as softening substance, the hardness of water seems to soften most members’ water samples by half according to the drops of EDTA except Adam Holyk’s water sample. Adam Holyk’s water sample does not change much after adding the baking soda. His Original water sample is five drops of EDTA, but after adding the baking soda, it only decreases one drop of EDTA. Bo-Yang Chen’s water sample is four drops of EDTA, which original water sample is nine drops of EDTA. Carol Chen’s water sample is two drops of EDTA, which original water sample is five drops of EDTA. And, Jason Cillo’s water sample is two drops of EDTA, which original water sample is four drops of EDTA.
My hypothesis is that the water samples from PA should have the highest hardness of water. However, the experiment data does not support it. From the experiments and tests, people can suggest Pennsylvania and Jew Jersey have a lower hardness of water, which California has the highest hardness of water. The main reason can be that both State College and New Jersey are famous for agriculture. Moreover, the quality of water will be examined more seriously than California. Furthermore, my hypothesis was saying longer path creates higher hardness of water because water will absorb the mineral and ions from stones and rocks. In this case, California should have the highest hardness of water because California is approximately four times larger than Pennsylvania and twenty times larger than New Jersey.
To compare the results from AA and EDTA, from Bo-Yang Chen’s water sample, it has 122 ppm of CaCO3 in the AA test and 180 ppm of CaCO3 in the EDTA test. The percent error is 47.5% if we are assuming the AA test is correct and the EDTA test is incorrect. The AA test should be a better method of testing water hardness since it contains less manual operations. Furthermore, the EDTA test can be less accurate because EDTA reacts with all divalent cations, not just Ca2+, and Mg2+. This means other ions in the water sample can influence the result while the AA test would not be influenced by other ions.
Conclusion
In this experiment, the idea was testing a water sample and determine the hardness of the sample by calculating the parts per million (ppm) of Calcium, Magnesium ions, and CaCO3 in the samples. AA and EDTA tests are the two methods to determine water hardness. Both of the methods show the hardness of water samples, but the data were different. Additionally, in the experiment, Bo-Yang Chen’s water sample from California has the highest hardness of water, which directly conflict with my hypothesis that Pennsylvania would have the highest hardness of water. The main reason can be Pennsylvania is known for its agriculture. Therefore, the quality of water is better than California where water probably goes through longer paths.
 The Chemistry of Natural Waters
The Chemistry of Natural Waters 






